Research
Research in the Wozniak lab focuses on innate immune cell interactions with the fungal pathogen Cryptococcus neoformans.
Current Research: We work on three main projects in the lab: 1) examining the roles of differentially-regulated genes in antifungal activity by macrophage and dendritic cell subsets, 2) examining the mechanism(s) involved in antfungal activity by lysosomal enzymes from DCs, and 3) examining antifungal potential of novel antifungal compounds.
Cryptococcus neoformans is an opportunistic fungal pathogen that primarily affects immune compromised patients, including those with AIDS and those on immune suppressive therapies to prevent organ transplant rejection. The disease begins as a pulmonary infection that eventually spreads to the central nervous system causing meningitis. Current estimates suggest that approximately 152,000 people are infected with this pathogen each year, and approximately 112,000 die each year due to cryptococcal meningitis (Rajasingham et al, The Lancet, 2022). The initial interaction with the host begins in the lung, and the innate immune cells of the lung (primarily macrophages and dendritic cells) are the front-line of defense against this pathogen.
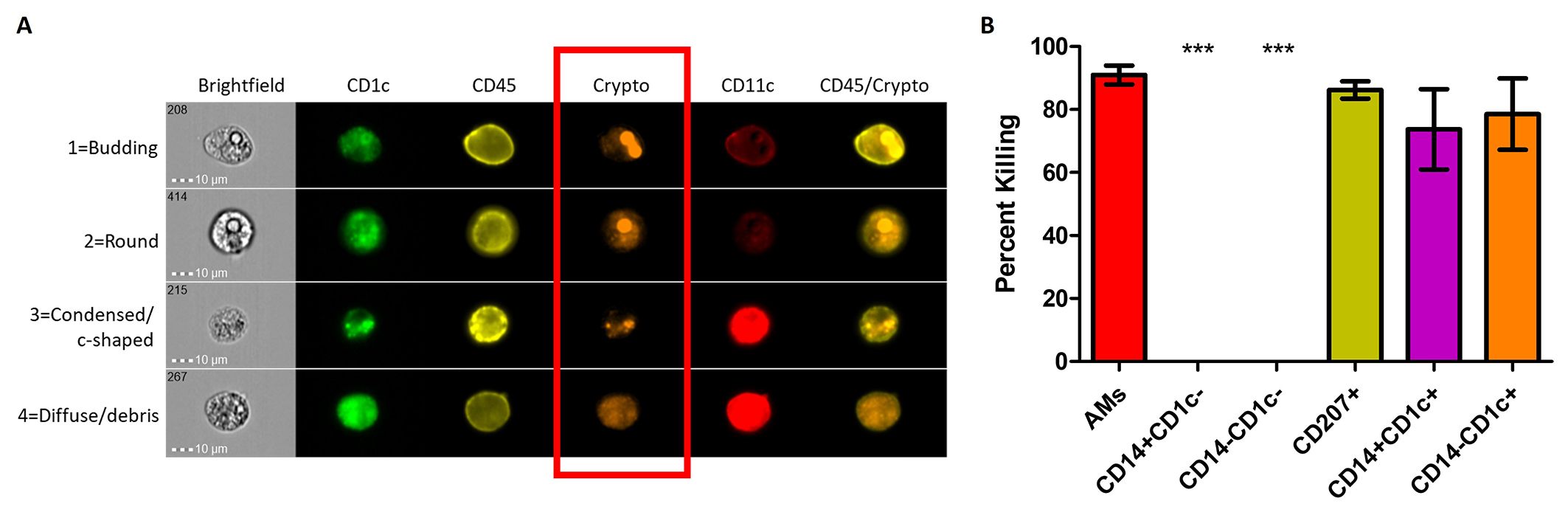
Role of differentially-regulated genes in antifungal activity of macrophage and DC subsets: This project examines the macrophage and DC side of the interaction of fungal pathogens, including C. neoformans, with different subsets of these phagocytes. Many laboratories have examined intracellular growth of C. neoformans and other fungal pathogens inside of macrophages, primarily focusing on cell lines. In primary cells from mice and humans, macrophages subsets exist that interact differently with pathogens.We have shown that the interaction of C. neofomans with two different subsets of human macrophages results in two different outcomes – intracellular growth or intracellular killing. RNA-seq data revealed that the most differentially-regulated genes included antigen processing genes and metabolic genes. We are currently examining the roles of these differentially-regulated genes in antifungal activity. These studies can also be applied to other fungal pathogens. The ultimate goal of these studies is to identify mechanisms used by antifungal phagocytes that could be applied as immunotherapy against fatal C. neoformans infections.
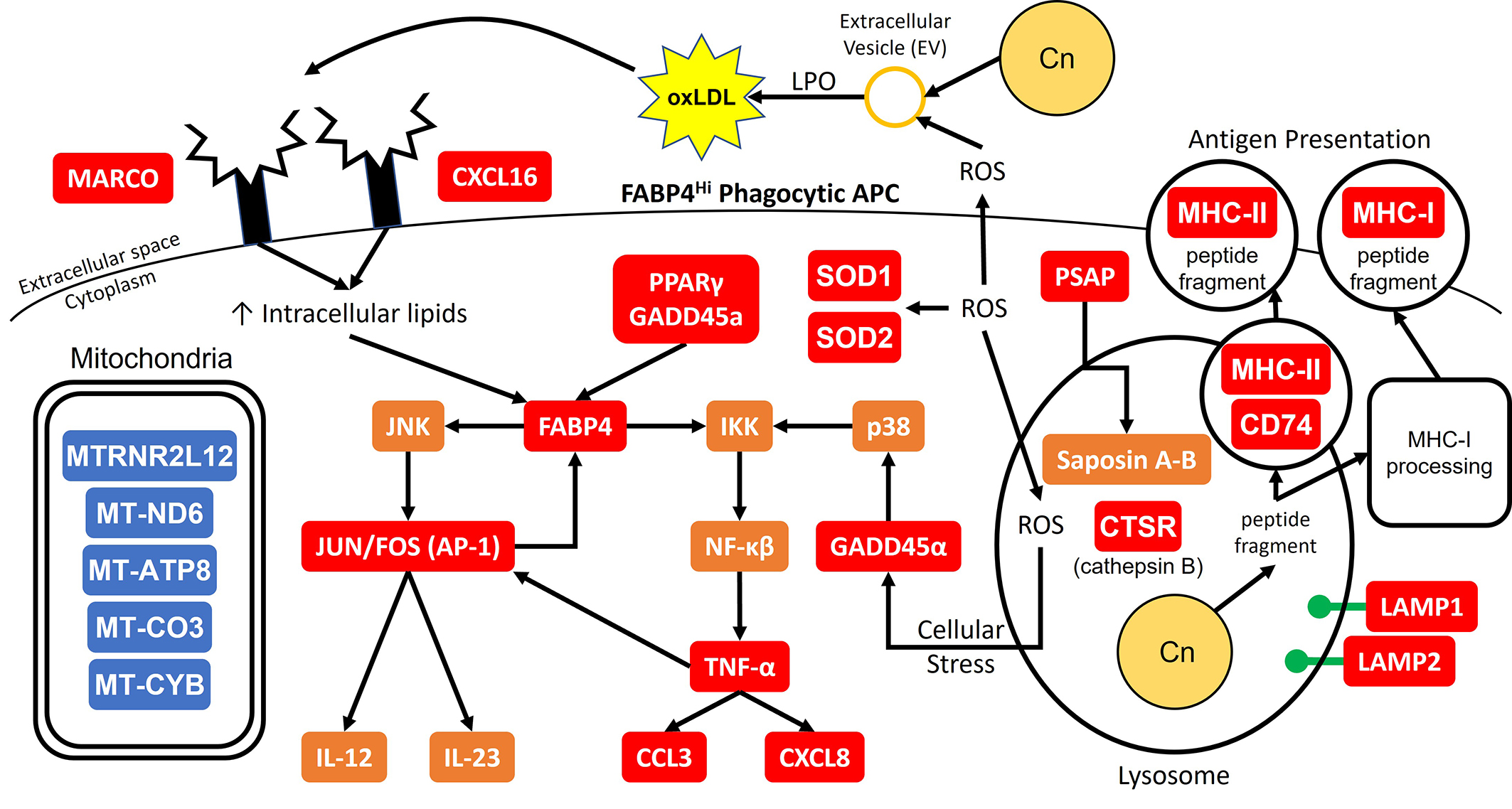
Mechanism(s) by which dendritic cell (DC) lysosomal enzymes have antifungal activity: This project examines mechanisms involved with lysosomal degradation of the cryptococcal organism.We have shown that the lysosomal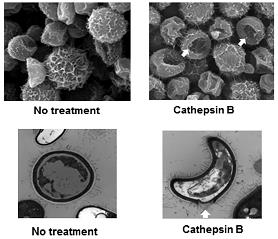 enzyme, cathepsin B, is capable of killing C. neoformans. This happens because of the formation of a hole in the cell wall and leads to osmotic lysis of the organism (Hole et. al, Scientific Reports, 2012). We also identified additional antifungal molecules from the DC lysosome (Nelson et al, Scientific Reports, 2021). We have identified 5 antifungal compounds found in the lysosome and are currently exploring their antifungal mechanisms using electron microscopy, fluorescent and confocal microscopy, and screening of cryptococcal mutant libraries.
enzyme, cathepsin B, is capable of killing C. neoformans. This happens because of the formation of a hole in the cell wall and leads to osmotic lysis of the organism (Hole et. al, Scientific Reports, 2012). We also identified additional antifungal molecules from the DC lysosome (Nelson et al, Scientific Reports, 2021). We have identified 5 antifungal compounds found in the lysosome and are currently exploring their antifungal mechanisms using electron microscopy, fluorescent and confocal microscopy, and screening of cryptococcal mutant libraries.
Mechanism(s) by which novel antifungal compounds have antifungal activity: These projects examine mechanisms involved antifungal mechanisms of novel antifungal molecules capable of killing C. neoformans. We have several collaborative studies ongoing in the lab, and we have found multiple anti-cryptococcal compounds (Gerasimchuk et al, Molecules, 2022 and unpublished observations). We are exploring the mechanisms using electron microscopy, fluorescent and confocal microscopy, and screening of cryptococcal mutant libraries.




